By: Lauren Commander | April 06, 2026 | 12 minutes read |
Share with each other
The presentation of the Miller School’s research on Alzheimer’s at this year’s AD/PD Conference highlights an important point about genetics, heredity and immunity.
Scientists from around the world gather every year at the International AD/PD Conference to share scientific advances and treatments for Alzheimer’s disease, Parkinson’s disease and related disorders. This year, the conference arrived in Copenhagen, and researchers from the University of Miami Miller School of Medicine arrived with a clear message: the field of Alzheimer’s continues to treat APOE4 not only as a major genetic risk but as a promising therapeutic target.
With nine oral presentations, the team at the Miller School of the John P. Hussman Institute for Human Genomics delivered one of the strongest single group presentations of the conference. Throughout the discussion, researchers followed new data on genetics and genetics that help improve the way APOE (especially APOE4)-related biology can be targeted in future treatments.
“APOE may be the main candidate we’re looking at for potential treatments right now,” said Margaret Pericak-Vance, Ph.D., director of the John P. Hussman Institute for Human Genomics and Dr. John T. Macdonald Foundation Professor of Human Genetics at the Miller School. “The APOE reports combine what we know about APOE and its protective effects, which identify high-risk populations. All of these things lead us to APOE as a treatment.”
From Amyloid to APOE: A Shift in Alzheimer’s Research
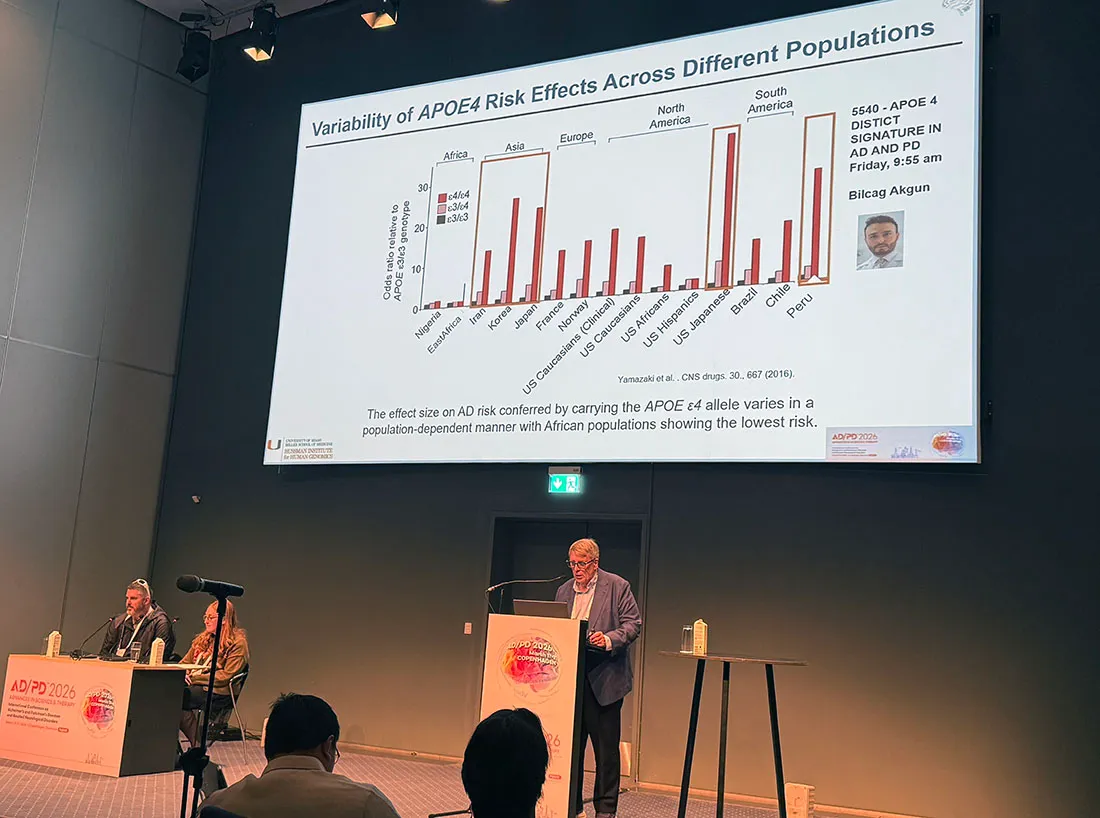
For many years, the biggest story focused on amyloid, while APOE4, discovered by Dr. Pericak-Vance in 1993, hiding in plain sight. Later ancestry mapping work in his lab helped clarify that the risk associated with APOE4 may be lower when the gene resides in an African background in a nearby genomic environment.
APOE has three common forms, APOE2, APOE3, and APOE4. APOE4 is heavy weight. One copy increases the risk significantly, and two copies almost guarantee Alzheimer’s disease if you live long enough. Scientists have debated whether APOE4 causes harm because it is toxic or dysfunctional. Jeffery Vance, MD, Ph.D., professor and founding chair of Dr. John T. Macdonald Foundation Department of Human Genetics, led a team of researchers that showed APOE4 is toxic and a strong candidate for treatment.
“APOE4 was covered for a long time by everyone who was pursuing amyloid,” Dr. Vance said. “So APOE4 is the target.”
How Genealogy Unlocks Nature’s Defense Against Alzheimer’s
The bottom line in all the statements is that, while APOE4 is the same protein across populations, risk is not.
“Same protein, different risks,” Dr. Vance said. “We have shown that the difference is driven by gene regulation, not changes in APOE4 itself.”
This turns genealogy into a scientific tool and suggests that personalized medicine may be the solution.
“If we are developing treatments, it may not be the same for everyone,” said Dr. Vance.
Mapping genes to new therapies
Together, the discussions followed a path from genetic discovery to mechanism to therapeutic logic.
One of the important results of the team includes a protective part of the gene that they identified about 2 million from APOE4 on African descendants. In people with two copies of APOE4, this region is associated with a 75 percent reduction in Alzheimer’s risk.
Dr Pericak-Vance said: “It’s a very important study.
Luciana Bertholm Nasciben, Ph.D., assistant scientist at the Miller School, used the forum to discuss her work using long-term DNA sequencing.
“Because this region is located in a complex region of the genome, I used long-term DNA sequencing to take a long period of DNA and reconstruct the region more accurately. We identified several unique elements that were inherited with a protective allele, suggesting that more research is needed to understand their importance and function,” said Dr. Bertholm Nasciben.
Dr. “Understanding this protective mutation can inform treatment options for all,” Vance said.
Several proposals have focused on ABCA7, a gene involved in lipid metabolism, because lipid metabolism affects how APOE works. Researchers at the Hussman Institute have previously shown that a 44 bp deletion, found only in African ancestry, is associated with Alzheimer’s disease, although some older carriers still know better. Several interviews focused on ABCA7.
YounJi Nam, Ph.D., assistant scientist at the Hussman Institute, discussed his findings that ABCA7 deletion disrupts the way neurons regulate essential fatty acids, especially ceramides. This may help explain its link to higher Alzheimer’s risk and suggest lipid pathways as a therapeutic target.
Assistant scientist Oladotun V. Olalusi, Ph.D., gave a speech showing that the known type of protection in ABCA7 has a strong protective effect when found in carriers of the deletion.
“The deleted protein is stable in the cell membrane and the protective SNP is located in the same part of the conserved protein. This suggests a strong protein interaction and may provide insight into the mechanism of how ABCA7 contributes to the risk of AD,” said Dr. Olalusi.

Modeling human brain cells
The Hussman Institute’s Aura Ramirez, Ph.D., described the use of adult blood cells from people with African, European and Peruvian ancestry to generate pluripotent stem cells (iPSC) and build complex types of cells such as the brain. When comparing control patterns across generations, the researchers also found evidence of early differentiation of brain tissue components among APOE4 carriers, suggesting that some changes begin early in life.
“It raises questions about when treatment should begin,” said Dr. Ramírez.
In a collaboration in Peru, Bilcag Akgun, MD, Ph.D., reported an increased risk of Alzheimer’s in Amerindian APOE4 carriers.
“This surprised us,” said Dr. Akgun, whose research team also discovered a rare mutation associated with Alzheimer’s risk that had previously been seen mainly in Asia. “These findings are interesting, since the risk of AD from APOE4 is highest in Asians, which means that the risk may have traveled from Asia through the Bering Strait during the migration of people to South America.”

Methylation markers in APOE4 carriers
In the MAGENTA project, Anthony J. Griswold, Ph.D., assistant professor in the Department of Society of Dr. John T. Macdonald of Human Genetics and co-director of the Hussman Institute Center for Genome Technology, and colleagues analyzed blood DNA methylation records from more than 600 people to more than 600 people who have studied Alzheimer’s worldwide.
“This analysis suggests that whole blood DNA methylation patterns may be specific indicators of Alzheimer’s,” said Dr. Griswold. However, there are still significant interactions of molecular pathways, which highlight the importance of multigenerational population studies for a comprehensive understanding of epigenetic risk for Alzheimer’s.
Effect of blood pressure on APOE4
Postdoctoral researcher Daniel Dorfsman, Ph.D., and Farid Rajabli, Ph.D., assistant professor of human genetics at the Miller School, highlighted a possible risk factor for AD – hypertension.
“APOEε4 risk is significantly exacerbated by the presence of hypertension, indicating an interaction rather than an additive effect,” they noted. “That is, the effect of the two together is greater than expected if one adds the individual risks.”
APOE3 has been used as a “neutral” allele to compare the protective effect of APOE2 with the risk associated with APOE4. Associate scientist Sofia Moura, Ph.D., reported her findings regarding the expression of APOE3 and APOE4 in different offspring.
“We found that the expression of APOE3 is really high in African tribes, which is different from the expression of APOE4, which is the lowest in African tribes,” said Dr. Moura. Therefore, the control of the two alleles does not work in the same way across generations.
Not all APOE4 risks are equal
Finally, Olabode E. Omotoso, senior research assistant, presented findings from the Hussman Institute led by DAWN Alzheimer’s Research, including data from 10 African countries and US partners. The findings suggest that the risk of Alzheimer’s associated with APOE4 is not uniform across Africa and appears to vary by region. East African groups showed the lowest perceived risk. West African groups have shown relatively high risk.
“This is exciting because it’s a big step towards a greater understanding of genetics across Africa and helps improve what we think we know about APOE4 involvement,” Dr. Pericak-Vance said.
As clinical trials for Alzheimer’s have historically failed at a rate close to 99 percent, researchers are looking to the effects of biological defenses as a way forward.
“Protective effects can be powerful tools,” said Dr. Vance. If something is naturally protective, it may have fewer side effects and work naturally. We just need to understand why they work.”
More from the Hussman Institute
Tags: Alzheimer’s disease, brain health, cognitive decline, Comprehensive Brain Health Center, Dr. James Galvin, Dr. Jeffery Vance, Dr. John T. Macdonald Foundation Department of Human Genetics, Dr. Margaret Pericak-Vance, genetics, genomics, Hussman Institute for Human Genomics, John P. Hussman Institute for Human Genomics
#Miller #School #International #Conference #Alzheimers #Parkinsons #Disease #InventUM
